 We will assume the expansion process is quasi-equilibrium, and the piston moves up an infinitesimal distance d. Upon receiving heat, the gas will tend to expand, pushing the piston up. The gas in the cylinder exerts an upward force, F=PA, where P is the gas pressure, and A is the cross-sectional area of the piston. Let us consider a piston-cylinder device, as illustrated in Figure 4.3.2. 
Work associated with the expansion and compression of a gas is commonly called boundary work because it is done at the boundary between a system and its surroundings. In equation form, the first law of thermodynamics is U Q W. In thermodynamics, an adiabatic process (Greek: adibatos. The first law of thermodynamics states that the change in internal energy of a closed system equals the net heat transfer into the system minus the net work done by the system. 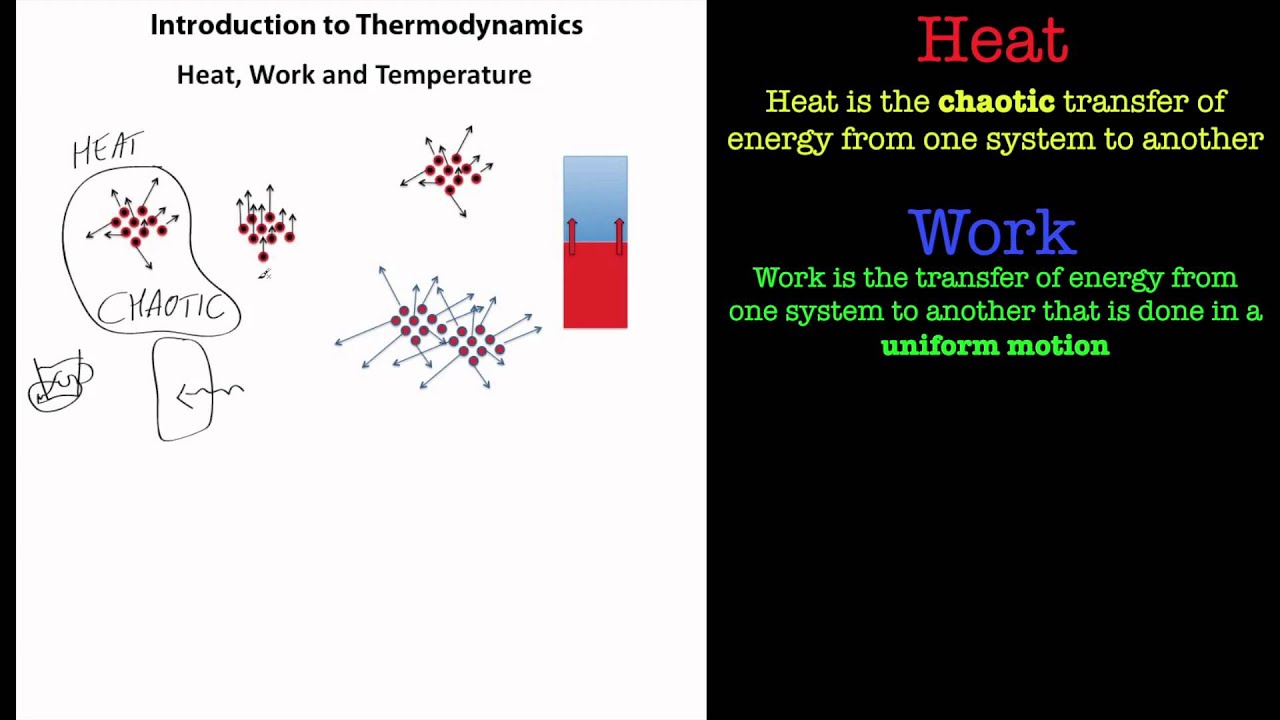
A piston-cylinder device initially contains 0.4kg og nitrogen gas at 160kPa and 140 degree Celcius. Figure 4.3.1 Work done due to a force acting on a block over a distance 4.3.1 Boundary work When an ideal gas is compressed adiabatically work is done on it and its temperature. Determine the boundary work done - Thermodynamics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
