 
Piece of cake, kinda like C – 12 on the left and 1 on the right. That means I’ll place a “12” in front of CO 2.Ģ K 4 + K 2Cr 2O 7 + H 2SO 4 → Fe 2(SO 4) 3 + Cr 2(SO 4) 3 + 12 CO 2 + H 2O + K 2SO 4 + KNO 3 
S looks tough since I see it in multiple terms on both sides. Great! I can do this….add a “2” on the left, in front of K 4, so now I have:Ģ K 4 + K 2Cr 2O 7 + H 2SO 4 → Fe 2(SO 4) 3 + Cr 2(SO 4) 3 + CO 2 + H 2O + K 2SO 4 + KNO 3.įe is balanced, for now. First question, how should I balance K? I know I’ll need to “double” the K counts on the right, but which term should I start with – K 2SO 4 or KNO 3? Not sure. Ok…what about on the right? Looks like a total 3 K on the right. I started with K since it’s the first atom I encounter from the left. How long do you think it will take to balance it? Which has 8 types of atoms in the equation: K, Fe, S, C, N, Cr, O and H. So, imagine if you have a real complex equation like this: K 4 + K 2Cr 2O 7 + H 2SO 4 → Fe 2(SO 4) 3 + Cr 2(SO 4) 3 + CO 2 + H 2O + K 2SO 4 + KNO 3 I mean, who wants to spend 30 minutes trying to balance ONE equation? Seriously, that 30 minutes is probably better-spent doing other things like watching YouTube, checking out latest posts in FB, Instagram, Twitter, uploading pictures to Snapchat, level up in that favorite game of yours or doing other things that will increase your happiness. There’s nothing wrong with that, but I think it gets a little daunting when it comes to having to keep track of multiple numbers of atoms (like 5 or more) and it will prolong the process unnecessarily. Most of the students I have worked with only knew Method 1 from their high school science/chemistry class. My quick google search seems to point towards Method 1 (by inspection or trial-error-method) and Method 2 (writing down atom counts) being the most popular methods when it comes to balancing equations. AsC l 3 aq + H 2 S aq → As 2 S 3 s + HCl aqĥ.Last week I posted on the three methods to balance chemical equations. If no coefficients are required to balance the equation, simply place the integer 1 in front of each reactant and/or product.Ĭlick "Check" to see whether your response below is the correct balanced chemical equation.ģ. Type integer coefficients in front of the reactants and products to balance the chemical equations. 
In the following examples, you will be given the chemical equations and states. Identify the states of matter: use the following subscripts for the compounds: (g) for gaseous substances, (s) for solids, (l) for liquids, and (aq) for aqueous solutions. Finally, write out the products on the right hand side of the equation using chemical nomenclature.īalance the equation: use the Law of Conservation of Mass to balance, or achieve the same number of atoms of every element on each side of the equation. The direction of the arrow indicates the flow of the reaction. Draw an arrow to represent "forms to react". Remember to use uppercase and lowercase letters, where appropriate. Write the unbalanced equation: using chemical nomenclature (see the Periodic Table of Elements ), write out the reactants on the left hand side of the equation. To balance chemical equations, the following three steps are used: Balancing a chemical equation is a mathematical approach used in chemistry to establish the relationship between the quantity of reactants and products. The equation shows the reactants on the left hand side and the products on the right hand side of the arrow. 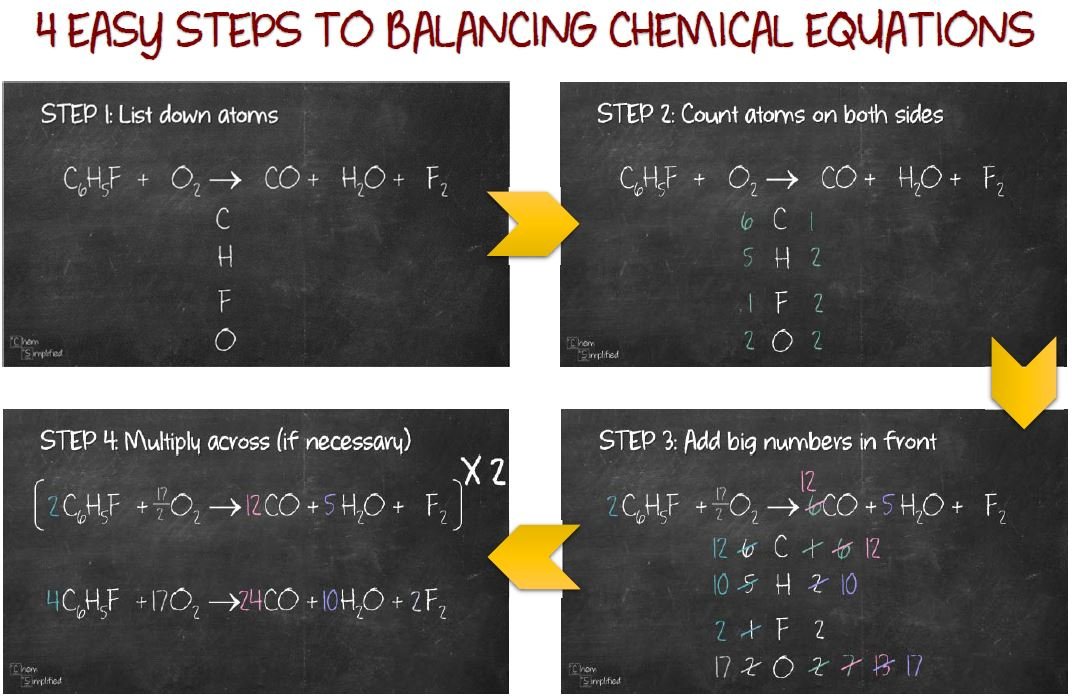
A chemical equation describes the changes that occur during a chemical reaction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
